Statins for primary prevention of cardiovascular disease in patients without diabetes
An evidence deep dive
Introduction
Some would say that statins should be in the water. Others would say they are a muscle-wasting poison. The truth is probably somewhere in between.
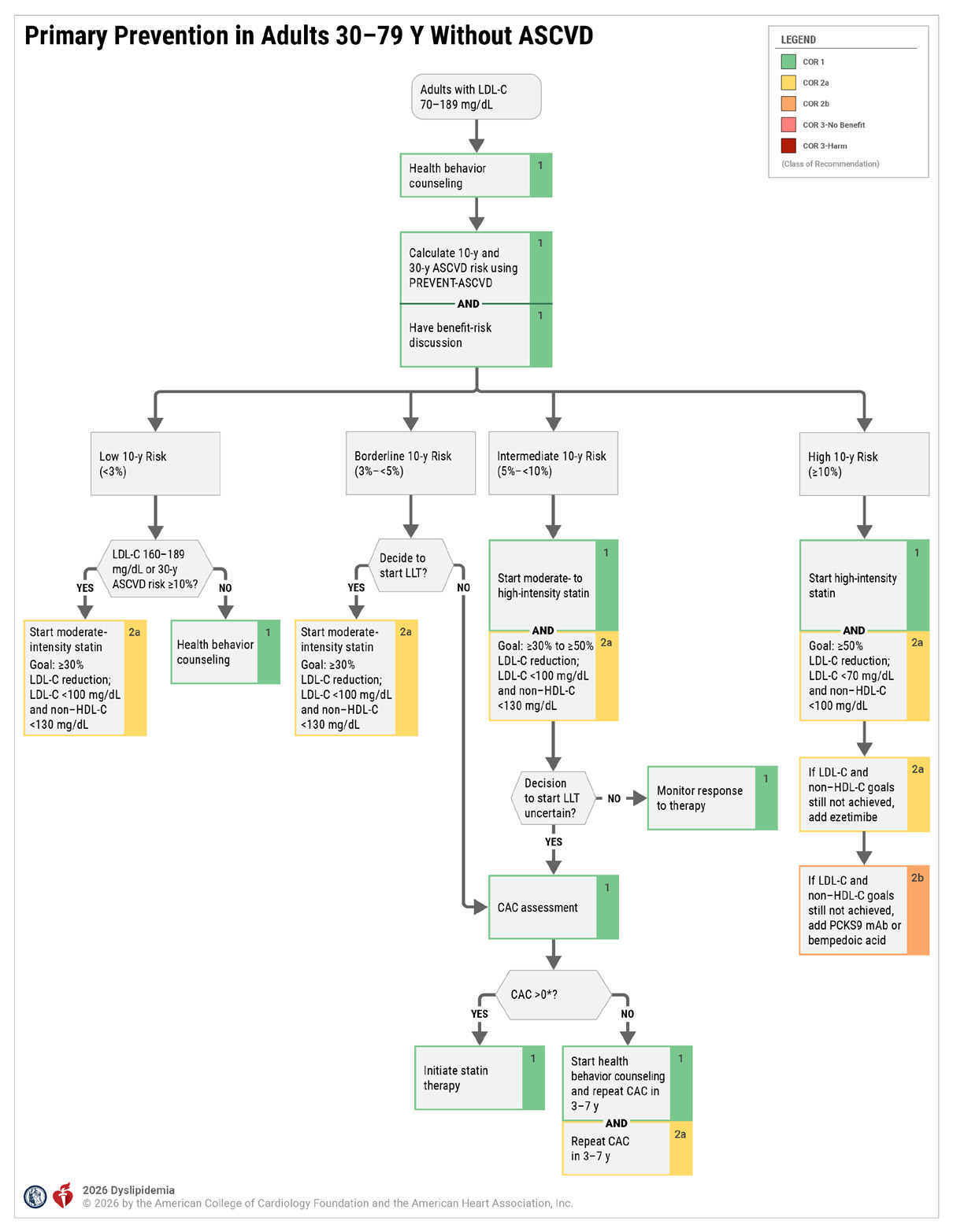
The ACC AHA 2026 Dyslipidemia guideline recommends using the PREVENT score to risk-stratify patients and offer the following flowchart to decide who should be on a statin (Blumenthal et al., 2026):
I wrote about the PREVENT score previously. It updates the PCE score by adding heart failure as an outcome and recalibrating risk using a contemporary cohort as the previous cohort overestimated the risk. The PREVENT score is roughly 5% lower than the PCE score for the same patient.
I played around with the PREVENT score a bit. If I set all the variables like blood pressure and cholesterol at the lowest end of normal and only adjusted the age, every man 55-79 years old and woman 59-79 years old would be recommended a statin. This is people with a systolic BP of 100, no diabetes, non-smokers, with a BMI of 20. That feels aggressive, which led me to this deep dive into the evidence underlying statins in low-risk patients. Based on the RCTs, which I explain below, I define low risk as: no diabetes, no history of cardiovascular disease, LDL <190, and normal CRP.
tldr
For a low-risk patient, I would not recommend a statin. I do not find convincing evidence for a benefit to all-cause mortality.
Efficacy from RCTs
For this deep dive I selected RCTs that evaluated statins for primary prevention in patients without diabetes and I focused on all-cause mortality as the patient-oriented outcome.
On one hand, avoiding a heart attack or stroke sounds good. On the other hand, we do not have clear evidence that avoiding cardiovascular events is a patient-oriented outcome. Thirty years ago, an MI was a death sentence and clearly a patient-oriented outcome. Today, with advances in medical technology and interventional cardiology, that is no longer the case. Modern meta-analyses have found that nonfatal MIs are not a good surrogate outcome for either cardiovascular or all-cause mortality (O’Fee et al., 2021). The authors speculate that this is due to the ability of high-sensitivity troponins to detect MIs with minimal myocardial injury and PCI technology to reverse the larger one’s.
Ultimately, I feel that any intervention for primary prevention needs to demonstrate an effect on all-cause mortality because there should be a high bar to take a person and turn them into a patient who takes a daily medication and needs lab monitoring.
Importantly, none of the individual RCTs were powered for all-cause mortality, so it is possible that a small improvement in all-cause mortality might go undetected due to low event rates. Accordingly, I spent more time thinking about meta-analyses for this question than I typically would.
ACAPS; Furberg et al., 1994
Population: 40-79 years old and mild cardiovascular disease defined by mild carotid artery stenosis. The average patient was 62 years old, white, and BP 131/77. 12% were smokers and 2% had diabetes. PREVENT score 8%.
Treatment: Lovastatin 20-40 mg (n = 460) vs placebo (n = 459) with goal LDL 90-110.
Follow-up: 2.8 years.
Change in LDL: 156 to 113 at 6 months.
Outcome: 88%!!! improvement in all-cause mortality and NNT 65. The mortality rate for the placebo group was 1.7%. There was also a 65% improvement and NNT 50 for cardiovascular events (MI, stroke, cardiovascular death). The cardiovascular event rate for the placebo group was 3.1%.
Safety: No statistically significant adverse events.
AFCAPS/TexCaps; Downs et al., 1998
Population: Men 45-73 years old and postmenopausal women 55-73 years old with LDL 130-190. The average patient was 58 years old, white, and BP 138/78. 12% were smokers and 2.5% had diabetes. PREVENT 7%.
Treatment: Lovastatin 20-40 mg (n = 3,304) vs placebo (n = 3,301) with goal LDL <110.
Follow-up: 5.2 years.
Change in LDL: 150 to 112 at 1 year.
Outcome: No difference in all-cause mortality. There was a 37% reduction and NNT 49 for acute major coronary events (fatal and nonfatal MI, unstable angina, sudden cardiac death). The event rate for the placebo group was 5.5%.
Safety: No statistically significant adverse events.
ALL-HAT, 2002
Population: >55 years old, hypertension, LDL 120-190. The average patient was 66 years old, white, and BP 145/84. 23% were smokers and 35% had diabetes. PREVENT 12%.
Treatment: Pravastatin 20-40 mg (n = 5,170) vs usual cares (n = 5,180) with goal LDL decrease of 25%. Of note, 25% of the usual cares patients were taking statins by the end of the study.
Follow-up: 4.8 years.
Change in LDL: 145 to 104 at end of study.
Outcome: No difference in all-cause mortality or fatal + nonfatal MI. The event rate for the placebo group was 8.1%.
Safety: No statistically significant adverse events.
ASCOT; Sever et al., 2003
Population: 40-79 years old, hypertension, total cholesterol <251. The average patient was 63 years old, white, and BP 164/95. 33% were smokers and 24% had diabetes. PREVENT 12%.
Treatment: Atorvastatin 10 mg (n = 5,168) vs usual cares (n = 5,137).
Follow-up: 3.3 years.
Change in LDL: 130 to 89 at end of study.
Outcome: No difference in all-cause mortality. There was a 36% reduction and NNT 90 for fatal + nonfatal MI. The event rate for the placebo group was 2.9%.
Safety: No statistically significant adverse events.
HOPE-3; Yusuf et al., 2016
Population: Men >55 years old, women > 65 years old, with one risk factor for cardiovascular disease. The average patient was 65 years old, Chinese, and BP 138/81. 27% were smokers and 5% had diabetes. PREVENT 10%.
Treatment: Rosuvastatin 10 mg (n = 6,361) vs usual cares (n = 6,344).
Follow-up: 5.6 years.
Change in LDL: 127 to 98 at end of study.
Outcome: No difference in all-cause mortality. There was a 24% reduction and NNT 90 for cardiovascular death and nonfatal MI or stroke. The event rate for the control group was 4.7%.
Safety: Increased muscle pain/weakness with rosuvastatin (5.8 vs 4.7%), increased cataract surgery with rosuvastatin (3.8 vs 3.1%).
JUPITER; Ridker et al., 2008
Population: Men >50 years old, women >60 years old, LDL <130, and CRP >2. The average patient was 66 years old, white, and BP 134/80. 15% were smokers and 0% had diabetes. PREVENT 9%.
Treatment: Rosuvastatin 20 mg (n = 8,901) vs placebo (n = 8,901).
Follow-up: 1.9 years.
Change in LDL: 108 to 55 at end of study.
Outcome: 20% reduction in all-cause mortality and NNT 181. There was a 44% reduction and NNT 81 for a composite cardiovascular outcome including fatal and nonfatal MI, fatal and nonfatal stroke, hospitalization for unstable angina, arterial revascularization, cardiovascular death. The event rate for the control group was 2.8%.
Safety: There was a 25% increase in new diabetes with NNH 166.
MEGA; Nakamura et al., 2006
Population: Men and postmenopausal women 40-70 years old, cholesterol 220-280. The average patient was 58 years old, Japanese, and BP 132/78. 15% were smokers and 0% had diabetes. PREVENT 9%.
Treatment: Pravastatin 10-20 mg (n = 3,966) vs usual cares (n = 3,866) with goal cholesterol <220.
Follow-up: 5.3 years.
Change in LDL: 157 to 122 at end of study.
Outcome: The p-value for all-cause mortality was 0.055 with a 28% risk reduction and NNT 90. There was a 33% reduction and NNT 119 for a composite cardiovascular outcome including fatal and nonfatal MI, angina, cardiac and sudden death, coronary revascularization. The event rate for the control group was 2.6%.
Safety: No statistically significant adverse events.
PROSPER; Shepherd et al., 2002
Population: 70-82 years old, hypertension, total cholesterol 154-348. The average patient was 75 years old, white, and BP 154/83. 27% were smokers and 11% had diabetes. PREVENT 20%.
Treatment: Pravastatin 40 mg (n = 2,891) vs placebo (n = 2,913).
Follow-up: 3.2 years.
Change in LDL: 147 to 107 at 2 years.
Outcome: No difference in all-cause mortality. There was a 15% reduction and NNT 47 for fatal + nonfatal MI and fatal + nonfatal stroke. The event rate for the placebo group was 16.2%.
Safety: There was a 25% increase in new cancer diagnoses in the pravastatin group with NNH 75. There was no difference when broken down to cancer subtypes, but the greatest effect was seen for GI cancers.
WOSCOPS; Shepherd et al., 1995
Population: Men 45-64 years old with cholesterol >252 and LDL >155 on two occasions with one occasion >174 and one occasion <232. The average patient was 55 years old, white, and BP 136/84. 44% were smokers and 1% had diabetes. PREVENT 6% but the average WOSCOPS patient would not qualify for PREVENT due to their baseline LDL of 192.
Treatment: Pravastatin 40 mg (n = 3,302) vs placebo (n = 3,293) with goal LDL <110.
Follow-up: 4.9 years.
Change in LDL: 192 to 142 at 6 months.
Outcome: The p-value for all-cause mortality was 0.051 with a 22% reduction and NNT 111. There was a 31% reduction and NNT 41 for fatal and nonfatal MI. The event rate for the placebo group was 7.5%.
Safety: No statistically significant adverse events.
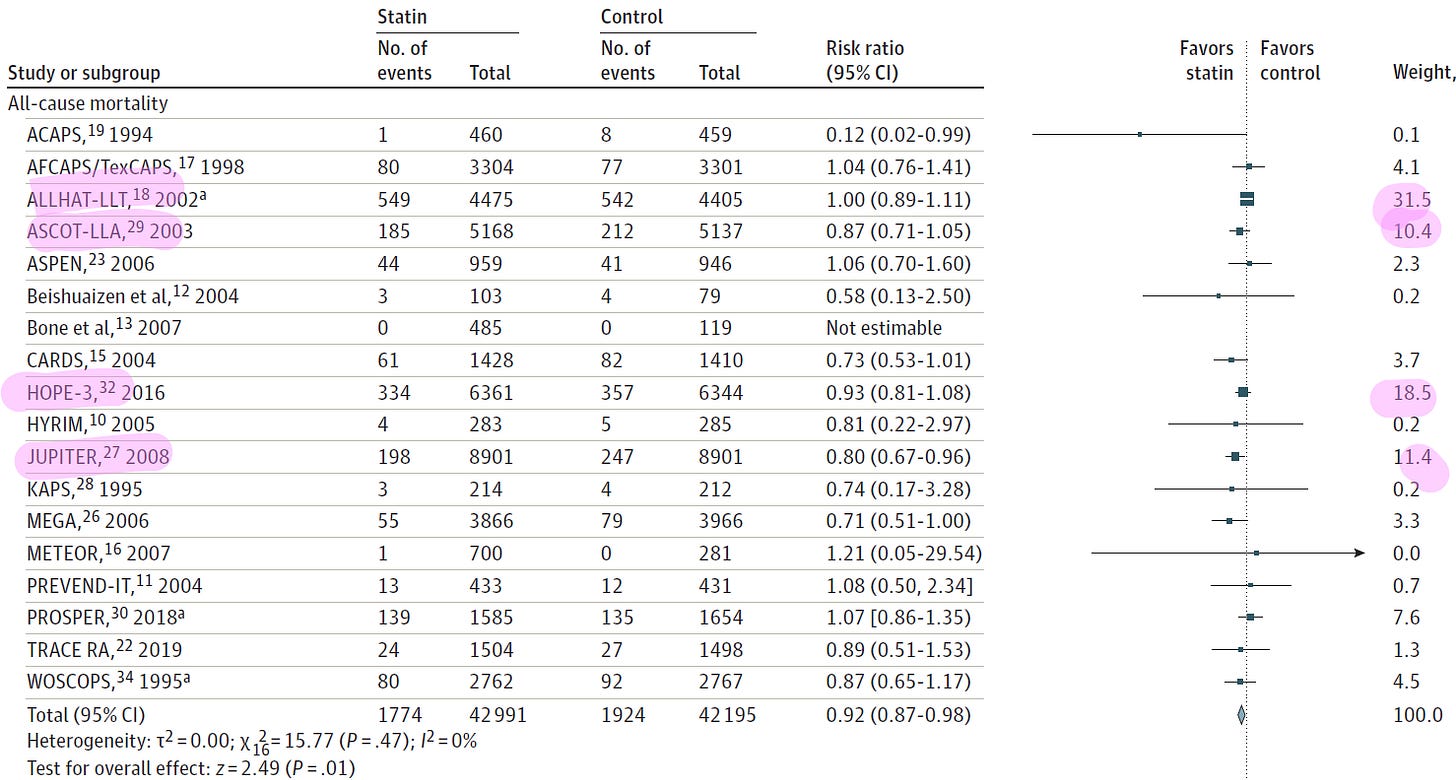
Before we move on to the meta-analyses I want to highlight three trials: ACAPS and JUPITER as the studies that found a statistically significant effect on all-cause mortality, and WOSCOPS as the study that was the closest to statistical significance.
For ACAPS, which found a striking 88% reduction in all-cause mortality and NNT 65, I tend to consider this as somewhat of a statistical anomaly. First, it was a small trial with a total of 919 patients. Second, AFCAPS enrolled a very similar population, used the same intervention, and found no effect on all-cause mortality.
For JUPITER, which found a 20% reduction in all-cause mortality and NNT 181, I do not consider this representative of a typical low-risk population. The premise of JUPITER was that inflammation is an independent risk marker for cardiovascular disease and it specifically studies a population of patients with normal LDL and elevated CRP.
The study that prompted JUPITER was a posthoc analysis of AFCAPS that found that lovastatin improved coronary event rates in patients with an above median LDL regardless of CRP, but that lovastatin only improved coronary event rates in patients with a below median LDL if they had an elevated CRP (Ridker et al., 2001). This would suggest that CRP could dictate a differential response to statins.
However, other studies like a subgroup analysis of HOPE-3 found no difference in effect size when comparing patients with normal vs elevated CRP. Similarly, a post-hoc analysis of JUPITER itself found that patients with higher CRP had higher rates of cardiovascular events but did not have a differential benefit to rosuvastatin when compared to lower CRP (Ridker et al., 2010).
It remains unclear whether CRP identifies a subgroup with a differential response to statins but considering that JUPITER only enrolled patients with an elevated CRP, I do not feel that it represents a typical low-risk patients.
The other caveat to JUPITER is that it was the only study that used a high-intensity statin. There is some evidence that high-intensity statins outperform moderate-intensity and my other conclusion from JUPITER is that we need an RCT of high-intensity statin for primary prevention in patients with a normal CRP (CTT, 2010).
WOSCOPS was the closest to finding a statistically significant effect on all-cause mortality (p for all-cause mortality was 0.051). However, the average patient in this trial had an LDL of 192, which would put patients in the severe hypercholesterolemia range. I do not think there is debate as to whether these patients should be on a statin and I do not find it reasonable to include this in an analysis of low-risk patients.
Efficacy from meta-analyses
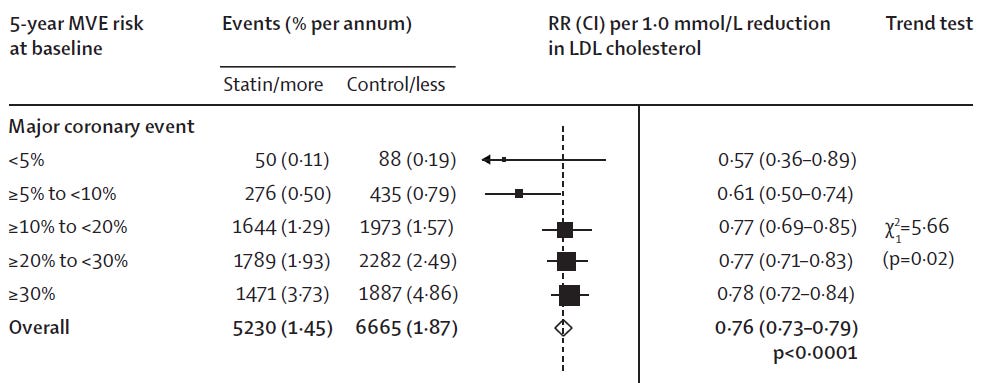
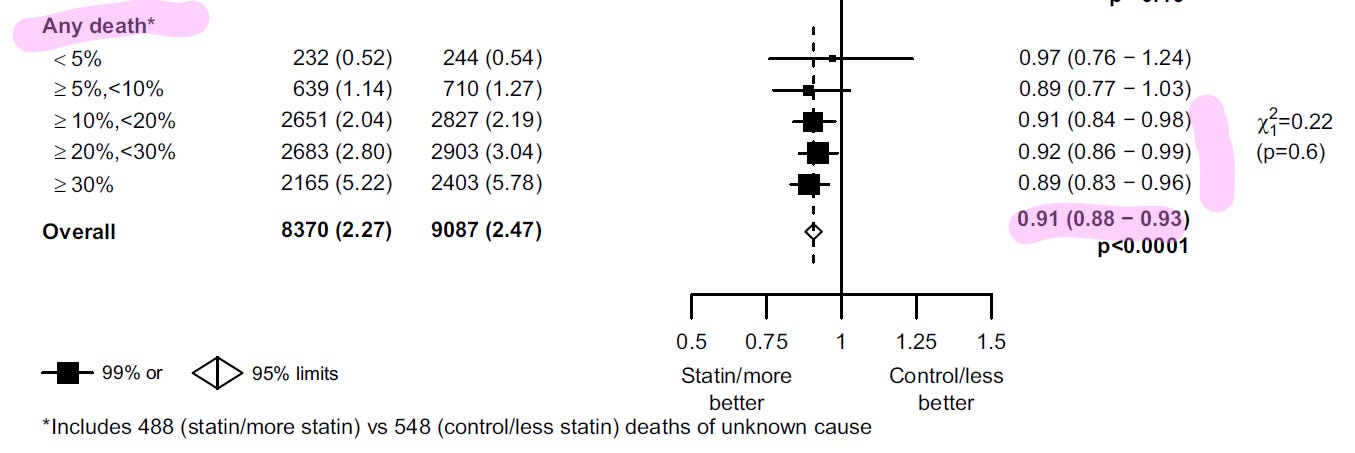
Cholesterol Treatment Trialist (CTT) collaboration, 2012
The CTT is a group of investigators that share their data to conduct individual patient-level meta-analyses. Having the individual patient-level data gives them more ability to converge on a precise outcome and also conduct more sophisticated subgroup analyses.
Sample: 27 RCTs which included both primary and secondary prevention studies.
Findings: They found that patients at lower risk had a greater relative risk reduction for major coronary events:
Interestingly, they did not find this trend for all-cause mortality and they did not observe a statistically significant effect on all-cause mortality until they reached patients with a >10% risk for having a vascular event in 5 years. I suppose this is because the younger patients that avoid a vascular event would not have died from it. In the whole population they found a 9% reduction in all-cause mortality with NNT 500.
USPSTF; Chou et al., 2022
Sample: 26 RCTs of primary prevention.
Findings: 8% reduction in all-cause mortality with NNT 286 at 1.6 years and a 28% reduction in composite cardiovascular events with NNT 78).
Ray et al., 2010
Sample: 9 RCTs of primary prevention in patients without diabetes.
Findings: No effect on all-cause mortality.
Meta-analyses can lead to different conclusions depending on how you determine the inclusion criteria. For this reason, I’m cautious about treating meta-analyses as the undisputed king of the evidence hierarchy. Of these three trials, I would consider Ray et al.’s study to be most representative of a low-risk patient, and this trial was negative for all-cause mortality.
Safety from meta-analyses
For safety-related findings, I find that meta-analyses are more useful because RCTs are almost never powered for safety outcomes. I tend to think of medication benefits as condition-specific, whereas medications harms are more generalizable. Thus, it’s not always reasonable to pool results to look for the evidence across heterogeneous patients but it’s more reasonable to pool results to look at safety.
The USPSTF meta-analysis found that statins were not associated with an increased risk of cancer, transaminitis, myalgia, rhabdomyolysis, or diabetes (Chou et al., 2022). They did note that one study on high-intensity statin did find statins caused diabetes. The CTT meta-analyses found that statins were associated with an increase in transaminitis (RR 1.41, NNH 1111; CTT, 2026) and diabetes (RR 1.10 for low/moderate-intensity and 1.36 for high-intensity, NNH 1000 and 76; CTT, 2024). They did not find an effect on myalgia.
Conclusions
Let’s start with statin safety. In the context of safety, statins are probably the most studied medication in all of human history. I don’t necessarily think this means they are safe, but I do think it means we have a really good idea of what side effects are caused by statins. For instance, there is lots of discussion about the muscle-related problems, but these are rare and much more likely to be due to the nocebo effect. There is a real effect on diabetes but mainly for high-intensity statins which would not typically be recommended for primary prevention.
Next, let’s use the USPSTF meta-analysis to think about efficacy (Chou et al., 2022). I want to start by highlighting the four studies that contributed the most to the all-cause mortality analysis.
One study (JUPITER) enrolled a specific population of patients with normal LDL and elevated CRP and I do not think this study should be generalized to the typical low-risk patient.
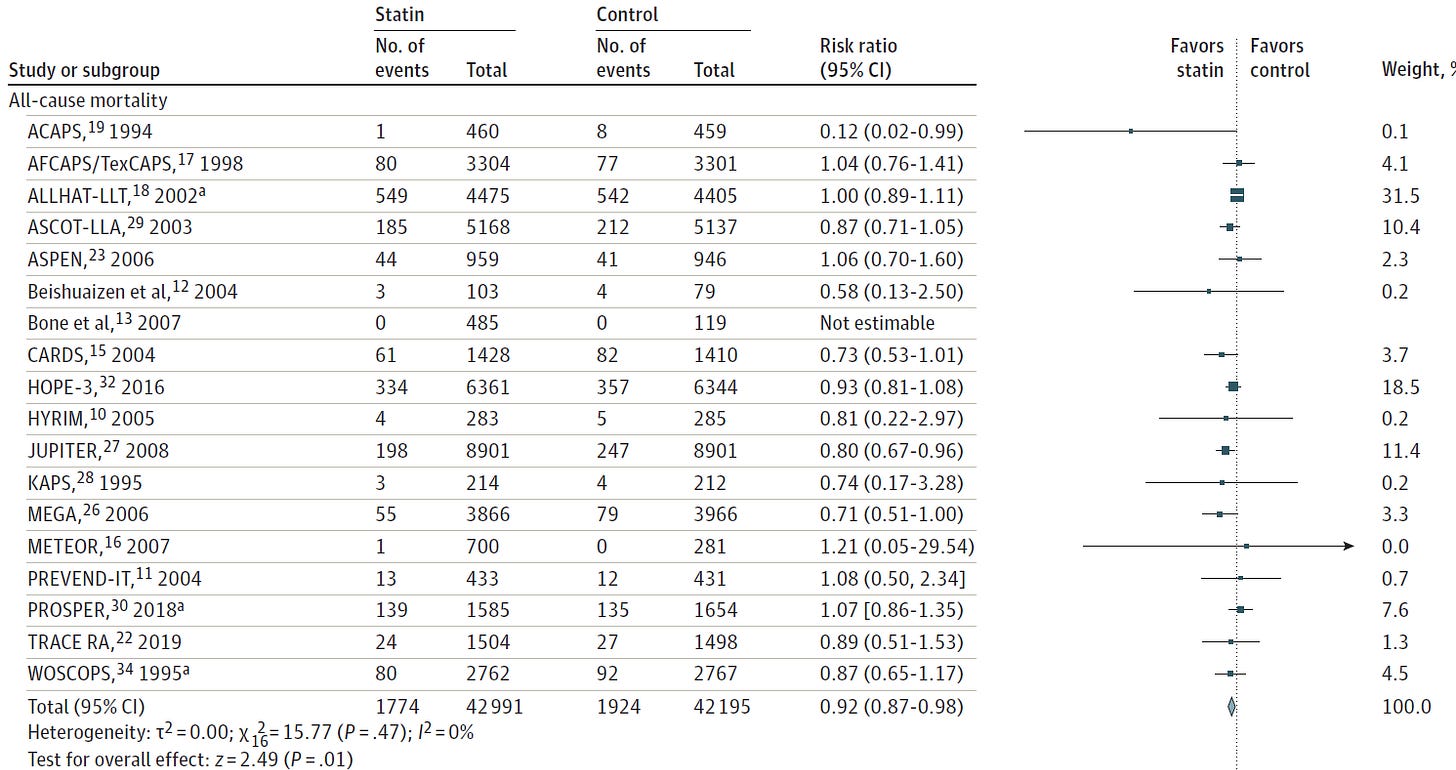
The other four studies (ALLHAT, ASCOT, HOPE-3) enrolled similar patients with high-normal to slightly elevated LDL that were treated with low or moderate-intensity statins. These three studies did not find an effect on all-cause mortality. In fact, the only primary prevention RCT powered for all-cause mortality was ALLHAT, which was stone cold negative with a risk ratio of 1.00.
Based on these studies, I do not think that statins improve all-cause mortality for the low-risk patient, which I would define as no diabetes, no history of cardiovascular disease, normal CRP, and LDL <190. Given the positive findings from JUPITER, I do think it is reasonable to check a CRP and consider statin treatment if the CRP is elevated.
A couple questions that this deep dive left me with:
Who should be on a statin? Second prevention? Diabetes?
Where is the line that a patient crosses over from low- to high-risk for primary prevention?
References
ALLHAT Officers and Coordinators for the ALLHAT Collaborative Research Group. The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial. Major outcomes in moderately hypercholesterolemic, hypertensive patients randomized to pravastatin vs usual care: The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT-LLT). JAMA. Dec 18 2002;288(23):2998–3007. doi:10.1001/jama.288.23.2998
Blumenthal RS, Morris PB, Gaudino M, et al. 2026 ACC/AHA/AACVPR/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA Guideline on the Management of Dyslipidemia: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. J Am Coll Cardiol. Mar 13 2026;doi:10.1016/j.jacc.2025.11.016
Cholesterol Treatment Trialists Collaboration. Assessment of adverse effects attributed to statin therapy in product labels: a meta-analysis of double-blind randomised controlled trials. Lancet. Feb 14 2026;407(10529):689–703. doi:10.1016/S0140-6736(25)01578-8
Cholesterol Treatment Trialists’ Collaboration. Effects of statin therapy on diagnoses of new-onset diabetes and worsening glycaemia in large-scale randomised blinded statin trials: an individual participant data meta-analysis. Lancet Diabetes Endocrinol. May 2024;12(5):306–319. doi:10.1016/S2213-8587(24)00040-8
Cholesterol Treatment Trialists C, Baigent C, Blackwell L, et al. Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet. Nov 13 2010;376(9753):1670–81. doi:10.1016/S0140-6736(10)61350-5
Cholesterol Treatment Trialists Collaboration, Mihaylova B, Emberson J, et al. The effects of lowering LDL cholesterol with statin therapy in people at low risk of vascular disease: meta-analysis of individual data from 27 randomised trials. Lancet. Aug 11 2012;380(9841):581–90. doi:10.1016/S0140-6736(12)60367-5
Chou R, Cantor A, Dana T, et al. Statin Use for the Primary Prevention of Cardiovascular Disease in Adults: Updated Evidence Report and Systematic Review for the US Preventive Services Task Force. JAMA. Aug 23 2022;328(8):754–771. doi:10.1001/jama.2022.12138
Downs JR, Clearfield M, Weis S, et al. Primary prevention of acute coronary events with lovastatin in men and women with average cholesterol levels: results of AFCAPS/TexCAPS. Air Force/Texas Coronary Atherosclerosis Prevention Study. JAMA. May 27 1998;279(20):1615–22. doi:10.1001/jama.279.20.1615
Furberg CD, Adams HP, Jr., Applegate WB, et al. Effect of lovastatin on early carotid atherosclerosis and cardiovascular events. Asymptomatic Carotid Artery Progression Study (ACAPS) Research Group. Circulation. Oct 1994;90(4):1679–87. doi:10.1161/01.cir.90.4.1679
Nakamura H, Arakawa K, Itakura H, et al. Primary prevention of cardiovascular disease with pravastatin in Japan (MEGA Study): a prospective randomised controlled trial. Lancet. Sep 30 2006;368(9542):1155–63. doi:10.1016/S0140-6736(06)69472-5
O’Fee K, Deych E, Ciani O, Brown DL. Assessment of Nonfatal Myocardial Infarction as a Surrogate for All-Cause and Cardiovascular Mortality in Treatment or Prevention of Coronary Artery Disease: A Meta-analysis of Randomized Clinical Trials. JAMA Intern Med. Dec 1 2021;181(12):1575–1587. doi:10.1001/jamainternmed.2021.5726
Ray KK, Seshasai SR, Erqou S, et al. Statins and all-cause mortality in high-risk primary prevention: a meta-analysis of 11 randomized controlled trials involving 65,229 participants. Arch Intern Med. Jun 28 2010;170(12):1024–31. doi:10.1001/archinternmed.2010.182
Ridker PM, Danielson E, Fonseca FA, et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N Engl J Med. Nov 20 2008;359(21):2195–207. doi:10.1056/NEJMoa0807646
Ridker PM, MacFadyen J, Libby P, Glynn RJ. Relation of baseline high-sensitivity C-reactive protein level to cardiovascular outcomes with rosuvastatin in the Justification for Use of statins in Prevention: an Intervention Trial Evaluating Rosuvastatin (JUPITER). Am J Cardiol. Jul 15 2010;106(2):204–9. doi:10.1016/j.amjcard.2010.03.018
Ridker PM, Rifai N, Clearfield M, et al. Measurement of C-reactive protein for the targeting of statin therapy in the primary prevention of acute coronary events. N Engl J Med. Jun 28 2001;344(26):1959–65. doi:10.1056/NEJM200106283442601
Sever PS, Dahlof B, Poulter NR, et al. Prevention of coronary and stroke events with atorvastatin in hypertensive patients who have average or lower-than-average cholesterol concentrations, in the Anglo-Scandinavian Cardiac Outcomes Trial--Lipid Lowering Arm (ASCOT-LLA): a multicentre randomised controlled trial. Lancet. Apr 5 2003;361(9364):1149–58. doi:10.1016/S0140-6736(03)12948-0
Shepherd J, Blauw GJ, Murphy MB, et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): a randomised controlled trial. Lancet. Nov 23 2002;360(9346):1623–30. doi:10.1016/s0140-6736(02)11600-x
Shepherd J, Cobbe SM, Ford I, et al. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. West of Scotland Coronary Prevention Study Group. N Engl J Med. Nov 16 1995;333(20):1301–7. doi:10.1056/NEJM199511163332001
Yusuf S, Bosch J, Dagenais G, et al. Cholesterol Lowering in Intermediate-Risk Persons without Cardiovascular Disease. N Engl J Med. May 26 2016;374(21):2021–31. doi:10.1056/NEJMoa1600176

Would you say that the PREVENT score may overestimate risk then based on the type of recommendation it made for low risk patients? Also, how well does the scoring tool apply to other demographics?